LB4. Patterns and Predictors of Recurrence Following Neoadjuvant Chemoimmunotherapy and Surgery in Non-Small Cell Lung Cancer
Objective: Neoadjuvant chemoimmunotherapy (chemoIO) is standard of care for locally advanced, resectable NSCLC without targetable EGFR/ALK alterations. However, little is known regarding predictors of recurrence and recurrence patterns after surgery. To address this knowledge gap, we investigated clinicopathologic risk factors associated with recurrence following neoadjuvant chemoIO and surgery in this patient population.
Methods: This is a multicenter, international, retrospective cohort study of patients with clinical stage IA3-III NSCLC without known targetable EGFR/ALK alterations who underwent neoadjuvant chemoIO and surgical resection from 1/2017-3/2024. Exclusion criteria were prior history of lung cancer, treatment with investigational immunotherapies or other neoadjuvant treatments. The primary endpoint was recurrence-free survival (RFS), which was measured from date of surgery and estimated using the Kaplan-Meier approach with Cox proportional hazards models for univariable and multivariable analysis.
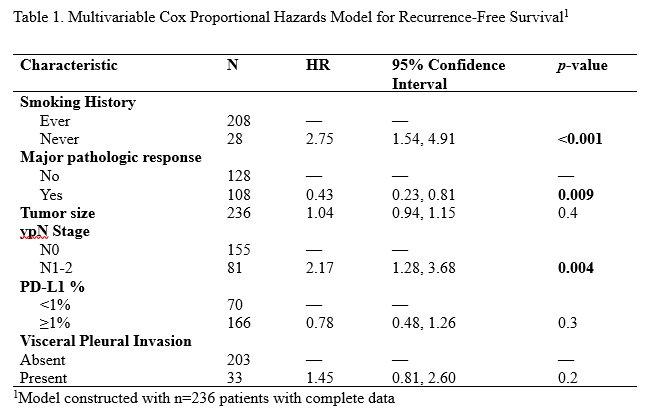
Results: 291 patients with a median follow-up of 29.3 months (95% CI 27.6-31.5 months) were included. Patients were treated with regimens containing anti-PD-(L)1 inhibitors in a neoadjuvant-only (247, 85%) or perioperative (44, 15%) approach. Clinical N2 disease was present in 119 (41%) patients. R0 resection was achieved in 281 (97%) cases and 182 (63%) cases were completed minimally-invasively. A major pathologic response (MPR) was seen in 141 (48%) cases and a pathologic complete response (pCR) was seen in 84 (29%) cases. Median residual viable tumor (RVT) was 10% (IQR 0-60%). Following surgery, 72 (25%) patients received adjuvant therapy. Of these, 14 (19%) received post-operative radiation (PORT) alone, 45 (63%) received systemic therapy alone, and 13 (18%) received a combination of systemic therapy and PORT. Systemic therapy regimens included chemotherapy alone (13/72, 18%), immunotherapy alone (37/72, 51%), and chemoIO (7/72, 10%) for a median of 4.5 cycles (range 1-14). A total of 90 recurrences were observed during the study period, of which 35 (39%) were locoregional, 36 (40%) were distant, and 19 (21%) were both locoregional and distant. The most common site of locoregional recurrence was lymph nodes (33/90, 37%). The most common site of distant recurrence was the brain (19/90, 21%). In patients with a pCR, 9/84 (11%) experienced recurrences, with 5/9 (56%) locoregional recurrences in lymph nodes. Seven patients with 90-day postoperative mortalities and 10 patients with R1 resections were excluded from RFS analyses. The 2-year RFS rate was 71.6% (95% CI 66.2-77.4%). On univariable analysis, never smoking status, cN2 disease , adenocarcinoma histology, increasing primary tumor invasive size, increasing ypN stage, increasing RVT%, absence of MPR, absence of pCR, lymphovascular invasion, visceral pleural invasion, spread through airspaces, and PD-L1<1% were associated with worse RFS (p<0.05). On multivariable analysis, never smoking status, absence of MPR and residual nodal disease independently predicted worse RFS (Table 1).
Conclusions: Despite high rates of MPR and pCR, recurrence occurred in nearly one-third of patients by two years after surgery. Never smoking status, absence of MPR, and residual nodal disease were independent predictors of recurrence. These findings support prospective evaluation of risk-adapted postoperative treatment strategies, including surveillance, in this patient population.
|