马上注册,阅读更多内容,享用更多功能!
您需要 登录 才可以下载或查看,没有账号?立即注册 
×
Jones DR, Opitz I, Harpole D, Yanagawa J, Lim E, Tsutani Y, Tan DSW, Dacic S, Ganti AK, Bodla S, Batig A, Lyfar P, Forcina A, Felip E. TROPION-Lung12: A phase 3 study of adjuvant datopotamab deruxtecan and rilvegostomig in ctDNA-positive or high-risk pathology stage I non-small cell lung cancer. J Thorac Cardiovasc Surg. 2026 Jan;171(1):1-9. doi: 10.1016/j.jtcvs.2025.09.017. Epub 2025 Sep 19. PMID: 40976544; PMCID: PMC12536336.
TROPION-Lung12: A phase 3 study of adjuvant datopotamab deruxtecan and rilvegostomig in ctDNA-positive or high-risk pathology stage I non-small cell lung cancer - PubMed
(OA)TROPION-Lung12: A phase 3 study of adjuvant datopotamab deruxtecan and rilvegostomig in ctDNA-positive or high-risk pathology stage I non–small cell lung cancer - The Journal of Thoracic and Cardiovascular Surgery
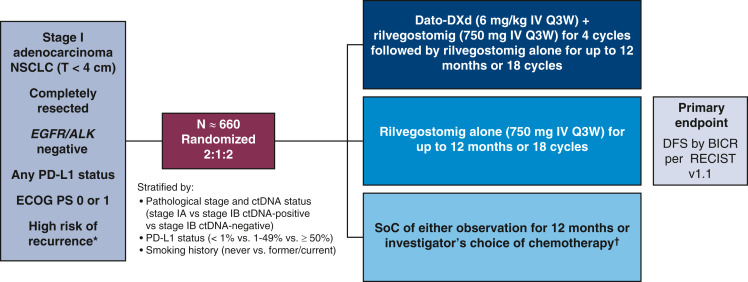
TROPION-Lung12 (NCT06564844) is a phase 3 randomized study enrolling approximately 660 patients with stage I adenocarcinoma without actionable genomic alterations who have undergone complete surgical resection. Eligible patients must have preoperative ctDNA-positive status or 1 or more high-risk pathologic features. Patients will be randomized 2:1:2 to receive Dato-DXd (6 mg/kg intravenously [IV] every 3 weeks) plus rilvegostomig (750 mg IV every 3 weeks) for 4 cycles, followed by rilvegostomig (up to 12 months/18 cycles total), rilvegostomig alone (up to 12 months/18 cycles total), or standard of care (SoC) for up to 12 months. The primary endpoint is disease-free survival, assessed using blinded independent central review according to RECIST v1.1, with key secondary endpoints including overall survival, for the Dato-DXd plus rilvegostomig arm versus the SoC arm.
德达博妥单抗(Dato-DXd,datopotamab deruxtecan,DS-1062a)
Rilvegostomig 是一款靶向 PD-1/TIGIT 双抗,全球和国内的最高研究进度均为 III 期临床阶段。阿斯利康针对 Rilvegostomig 布局了多个适应症,其中胆道癌、非小细胞肺癌、胃癌均已进入临床 III 期。
|
|